Xanthate is one of the most commonly used collectors in flotation of sulfide ores. To ensure its full effectiveness, the following aspects should be considered during use and storage:
(1) Use it in an alkaline pulp whenever possible. Xanthate is easily dissociated in water, leading to hydrolysis and decomposition. For example, the reaction formula for sodium xanthate is as follows:
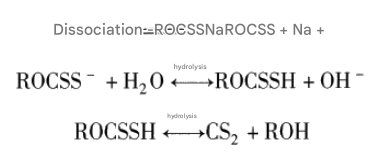
Dissociation of xanthate is desirable because the collecting action is mainly due to the xanthate anion (ROCSS-). However, if left uncontrolled, the reaction will hydrolyze into xanthic acid (ROC-SSH), and eventually decompose into alcohol and carbon disulfide, becoming ineffective. From the above reaction, it can be seen that only by maintaining an alkaline pulp, i.e., increasing the mass fraction of OH, can the hydrolysis reaction proceed to the left, maintaining a higher mass fraction of xanthate anions and improving the collecting effect. Therefore, xanthate should generally be used in an alkaline pulp (pH greater than 7). If the xanthate needs to be used in acidic slurries, a higher grade xanthate should be used (because it decomposes more slowly than a lower grade in acidic slurries), and it should be added in multiple stages whenever possible.
(2) The xanthate solution should be prepared and used immediately. Because xanthate is easily hydrolyzed and decomposes, do not prepare too much at once. On-site, xanthate is generally prepared as a 1% aqueous solution. Hot water should never be used, as it will decompose faster when heated.
(3) To prevent xanthate decomposition and inactivation, it should be stored in a sealed container, away from humid air and water, in a dry, cool, and well-ventilated place. Avoid heat and take precautions against fire.