Cyanide is an important inhibitor in the flotation separation of non-ferrous metal sulfide ores. It is often used to inhibit sulfide minerals such as sphalerite, pyrite, and chalcopyrite. The commonly used cyanides in flotation are sodium cyanide and potassium cyanide, and sometimes ferricyanide is also used. Cyanide has a good inhibitory effect, but it has the following two major drawbacks:
(1) It is highly toxic. The following two points should be noted when using it:
1) It must be used in an alkaline pulp. After cyanide dissociates in water, it hydrolyzes to produce hydrogen cyanide. The reaction formula is as follows.
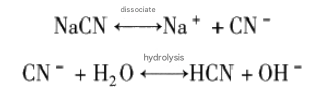
Hydrogen cyanide has a very low boiling point, only 26.5℃. It can volatilize into a highly toxic gas at room temperature, which seriously harms the human body. From the hydrolysis reaction, it can be seen that only when the pulp is alkaline will the equilibrium shift to the left, so that sodium cyanide exists in the CN- ion state, preventing the formation of hydrogen cyanide. Only when cyanide exists in the CN- state can it effectively exert its inhibitory effect.
2) The tailings water must be treated. (1) Treatment with oxidants (such as chlorine, bleaching powder, etc.) can be strengthened to decompose and deactivate cyanide in tailings water, achieving national discharge standards.
(2) Dissolving precious metals such as gold and silver can lead to the loss of these precious metals from the minerals.
Given these two drawbacks, cyanide should be used sparingly or avoided as much as possible in flotation. Sulfites, thiosulfates, zinc sulfate, etc., can be used to replace its inhibitory effect, but these substitutes are often less effective than cyanide.