

A certain ore body contains iron-copper ore in its central part and copper ore in the transitional zone at the edges. Its main metallic sulfide minerals are chalcopyrite, bornite, pyrite, and chalcocite; its main metallic oxide minerals are magnetite, hematite, native copper, and malachite. Minor minerals include marcasite, sphalerite, arsenopyrite, arsenite, azurite, cuprite, goethite, limonite, and specularite. Gangue minerals include calcite, dolomite, quartz, chalcedony, diopside, serpentine, kaolinite, chlorite, and sericite.
Due to the high copper and iron content, deep oxidation, and high mud content in the oxidized ore, coupled with a high copper binding rate, using a single flotation reagent consumes a large amount of reagent and has poor selectivity, making it difficult to obtain satisfactory results. Therefore, this ore adopted the sulfide flotation method, the process flow of which is shown in Figure 5-2.
Flotation reagents: Depressant: lime 1460 g/t (pH 8.5–9.5); Activator: sodium sulfide 563 g/t; Collector: xanthate 141 g/t; Frother: pine oil 59 g/t.
The ore is first sulfided at room temperature and then floated to obtain copper concentrate. Magnetic separation is used to separate the iron concentrate tailings. The specifications are: copper concentrate contains 18.10% copper; copper recovery rate 89.10%; iron concentrate contains 66.80% iron; iron recovery rate 65.30%.
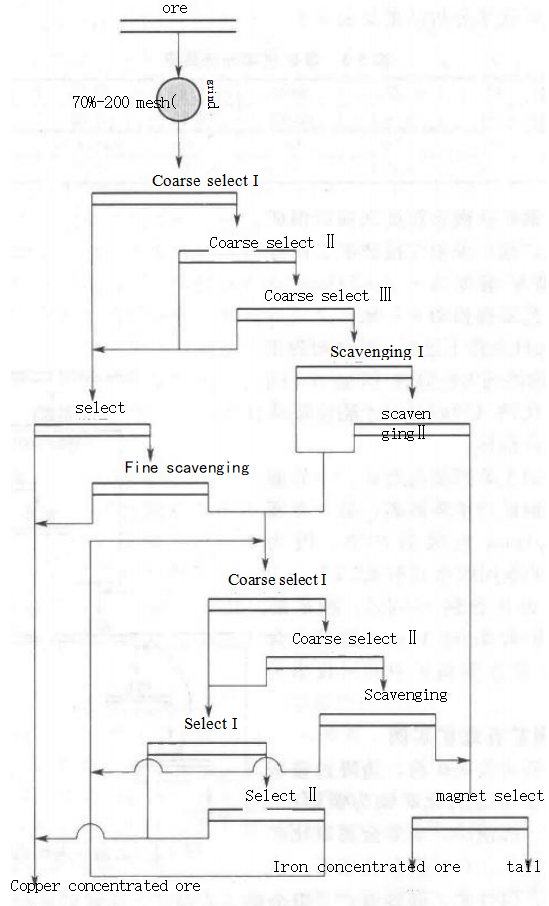
Figure 5-2 ore beneficiation process of a copper oxide ore beneficiation plant





